
In-depth Analysis of the Technological Innovation and Clinical Value of Three Devices
In late March, the technical team from Beijing WBL Medical Group visited the First Affiliated Hospital of Hainan Medical University to complete the installation, commissioning, and clinical training of three devices: the portable low-intensity pulsed ultrasound (LIPUS) device, the nerve sensitivity analyzer, and the extracorporeal pulsed ultrasound pelvic floor therapy platform. This marks the official launch of their collaboration in precision medicine, men’s health, and neurofunctional assessment.
Hospital Strength as Foundation, Innovative Technology as Empowerment

The First Affiliated Hospital of Hainan Medical University is a tertiary Grade-A comprehensive hospital, featuring eight national key clinical specialties and two key laboratories under the Ministry of Education. It serves as Hainan Province’s core medical-educational-research base. This training supports the hospital in upgrading its diagnostic and treatment capabilities in men’s health, neurofunctional assessment, and urological and andrological disciplines toward precision and non-invasive care. It fills a regional gap in non-invasive treatment for male pelvic floor dysfunction and enhances the quality of regional men’s health services.
Innovative Value and Clinical Applications of the Three Devices

Extracorporeal Pulsed Ultrasound Pelvic Floor Therapy Platform

This non-invasive device was specifically developed for male patients with pelvic floor dysfunction. Using extracorporeal pulsed ultrasound output, the energy penetrates the pelvic fascia to precisely target lesions in the prostate, pelvic nerves, and pelvic floor muscles. Through mechanical stress and thermal effects, it promotes local blood circulation, improves the microenvironment of pelvic floor tissues, activates the regenerative potential of damaged tissues, and restores normal contraction and regulatory function of pelvic floor muscles. Key clinical applications include chronic pelvic pain syndrome (CPPS), pelvic floor muscle dysfunction, voiding disorders with pelvic pain, and postoperative pelvic floor rehabilitation after prostate surgery. The device requires no surgical incisions or catheterization, achieving precise treatment through an external probe with minimal discomfort, substantially lowering the treatment threshold for male patients and improving adherence.
Nerve Sensitivity Analyzer

As a specialized instrument bridging basic and clinical medicine, this device employs a novel detection method controlled by computer to generate and record human responses to repeated thermal stimuli. It can accurately simulate and measure multi-dimensional sensory responses such as warm, cold, heat-induced pain, and cold-induced pain, with a core application in determining thermal pain thresholds. Widely applicable in both basic research and clinical diagnostics, the device provides important tools and objective, quantifiable data for the diagnosis, efficacy evaluation, and research of neurofunctional disorders. Its non-invasive, non-traumatic design ensures high safety and supports clinicians in addressing root causes, significantly reducing patient medical costs while providing broad medical benefits.
Portable Low-Intensity Pulsed Ultrasound Device (WBL-ED-BX)
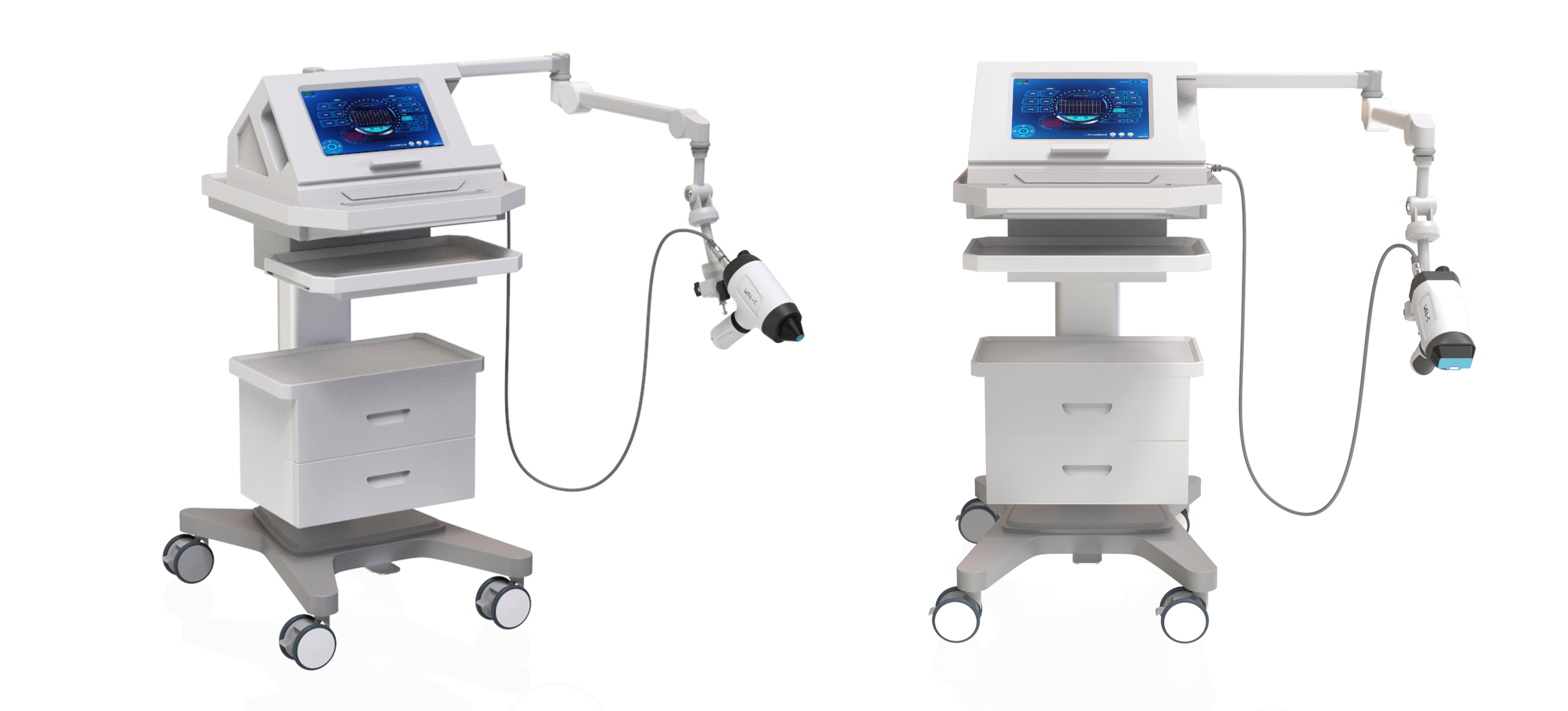
Based on the core technology of LIPUS, this device utilizes micro-mechanical and cavitation effects to precisely enhance blood flow in the penile corpora cavernosa, promote endothelial cell regeneration, and restore smooth muscle function. It offers a non-invasive clinical intervention and rehabilitation solution for patients with vascular or mixed erectile dysfunction (ED). The device is non-invasive, painless, and free of adverse effects such as hematoma. With its technical innovation and clinical practicality, it has won the National Medical Device Innovation Award and the IDEA International Design Award, becoming a benchmark product in the male ED treatment field.
Comprehensive Training to Ensure Standardized Clinical Application

To ensure the standardized and effective clinical deployment of the three innovative devices, WBL Medical implements a “theory + hands-on + long-term support” training system. The training covers four core modules: device principles, operational demonstrations, emergency response exercises, and maintenance protocols. Additionally, a 24-hour remote support group and scheduled maintenance guidance are established, creating a full-cycle technical support loop. After training, participants undergo both theoretical and practical assessments to ensure proficiency in all operational procedures, providing patients with standardized, safe, and efficient care. This marks the official clinical application of the three devices.

Medical-Industry Collaboration to Build a Healthy Ecosystem
WBL Medical adheres to a “medical-engineering integration” development model and has established deep cooperative relationships with multiple tertiary hospitals domestically and internationally. Its independently developed LIPUS series has received the IDEA International Design Award for its internationally advanced technology. The three newly launched core devices fill gaps in non-invasive treatment for male ED, quantitative assessment of neurofunction, and non-invasive intervention for male pelvic floor dysfunction. Moving forward, both parties will further advance clinical research and technological iteration, supporting comprehensive enhancement of regional men’s health services.

